 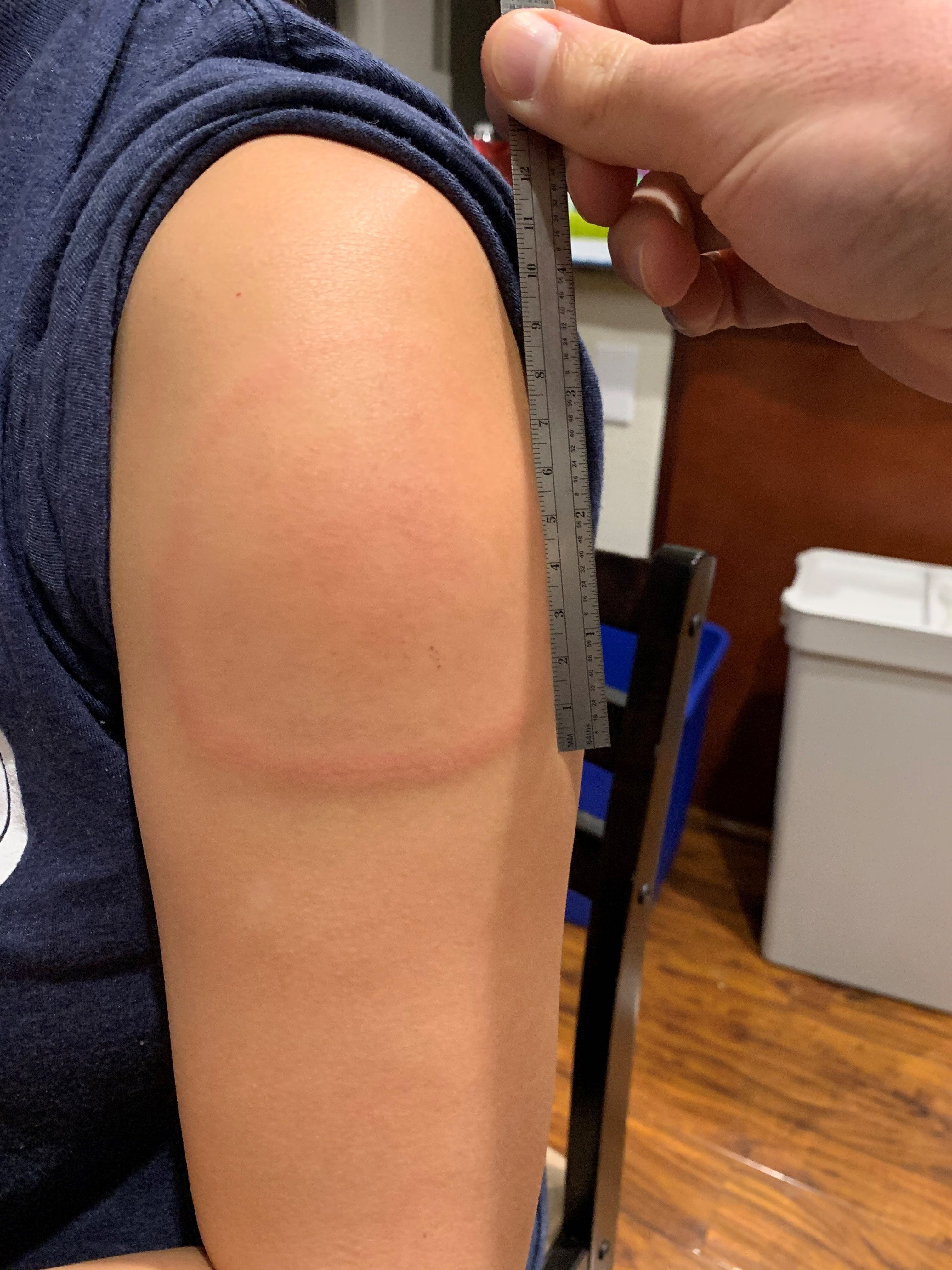
Very common (10% or more): Appetite loss (up to 32.1%) Musculoskeletal Very common (10% or more): Injection site pain (up to 97.2%), localized swelling/hardness (up to 17%), injection site erythema (up to 13.5%), localized swelling/induration (up to 12.6%)Ĭommon (1% to 10%): Delayed injection site reactions, injection site lymphadenopathyįrequency not reported: Injection site pruritus, injection site rash, injection site swelling, injection site urticaria ĭelayed injection site reactions included at least 1 of the following: erythema, pain, and/or swelling.įacial swelling occurred in 2 patients on Day 1 and Day 3 with a history of injection of dermatological fillers. Ĭommon (1% to 10%): Acute otitis media Local Postmarketing reports: Anaphylaxis ImmunologicĪcute otitis media occurred more frequently in patients 6 to 23 months of age who received this vaccine compared to placebo. Very common (10% or more): Nausea (up to 24%), vomiting (up to 24%) HematologicĬommon (1% to 10%): Lymphadenopathy HypersensitivityĬommon (1% to 10%): Hypersensitivity events/hypersensitivity Postmarketing reports: Acute urticaria, delayed urticaria, erythema multiforme Gastrointestinal Postmarketing reports: Myocarditis, pericarditis Dermatologic The most commonly reported side effects included injection site pain, headache, fatigue, and myalgia. Some dosage forms listed on this page may not apply to the brand name Moderna COVID-19 Bivalent Vaccine PF.Īpplies to sars-cov-2 (covid-19) mrna-1273 (bivalent booster) vaccine: intramuscular suspension. 
Note: This document contains side effect information about sars-cov-2 (covid-19) mrna-1273 (bivalent booster) vaccine. Generic name: sars-cov-2 (covid-19) mrna-1273 (bivalent booster) vaccine Moderna COVID-19 Bivalent Vaccine PF Side Effects 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
